BÜHLMANN fCAL® ELISA (calprotectin)

fecal Calprotectin (fCAL)- An FDA Cleared Gastrointestinal Test to Distinguish IBD from IBS
Calprotectin is a highly-sensitive clinical biomarker that has been shown to be extremely useful as an aid in diagnosis of inflammatory diseases in the gastrointestinal tract.
The BÜHLMANN fCAL® ELISA is an in vitro diagnostic assay intended for the quantitative measurement of fecal calprotectin in human stool. The BÜHLMANN fCAL® ELISA aids in the diagnosis of inflammatory bowel disease (IBD), specifically Crohn’s disease (CD) and ulcerative colitis (UC) and aids in the differentiation of IBD from irritable bowel syndrome (IBS) in conjunction with other laboratory and clinical findings.
This FDA 510k cleared, non-invasive, sensitive and specific assay is accurate, precise, efficient and offers reliable information to aid clinicians in selecting patients for further diagnostic procedures.
Product Information
| Name | BÜHLMANN fCAL® ELISA |
| Method | ELISA |
| Time to result | 2 h (approx.) |
| Sample type | 50-150 µl feces extract |
| Standard range | 30-1800 µg/g |
| Sensitivity (LoQ) | 30 µg/g |
| Instructions for Use & Other Downloads | see below |

CLINICIANS
Learn about the benefits of the BÜHLMANN fCAL® ELISA for your patients.

LABORATORY
Learn about the performance of the BÜHLMANN fCAL® ELISA
Learn More
The BÜHLMANN fCAL® ELISA sets a new standard in the industry:
| A clinical study with a total of 337 adult and pediatric patients was performed to assess the ability of the BÜHLMANN fCAL® ELISA to discriminate between patients with IBD and other non-inflammatory GI disorders, including IBS.
|
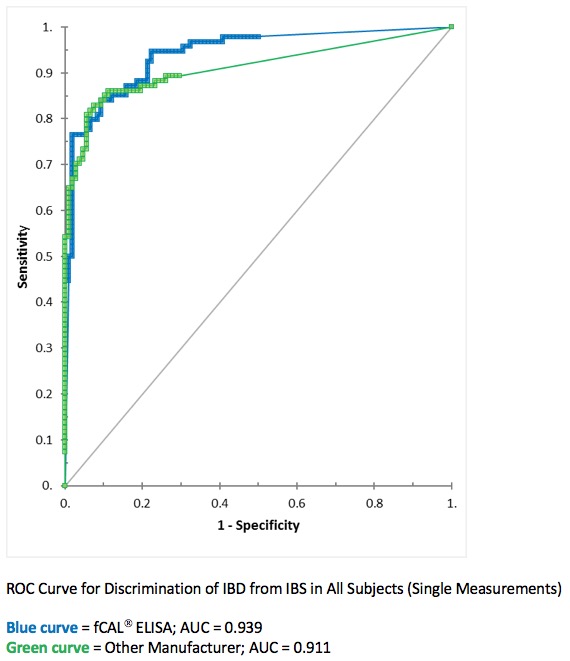 |
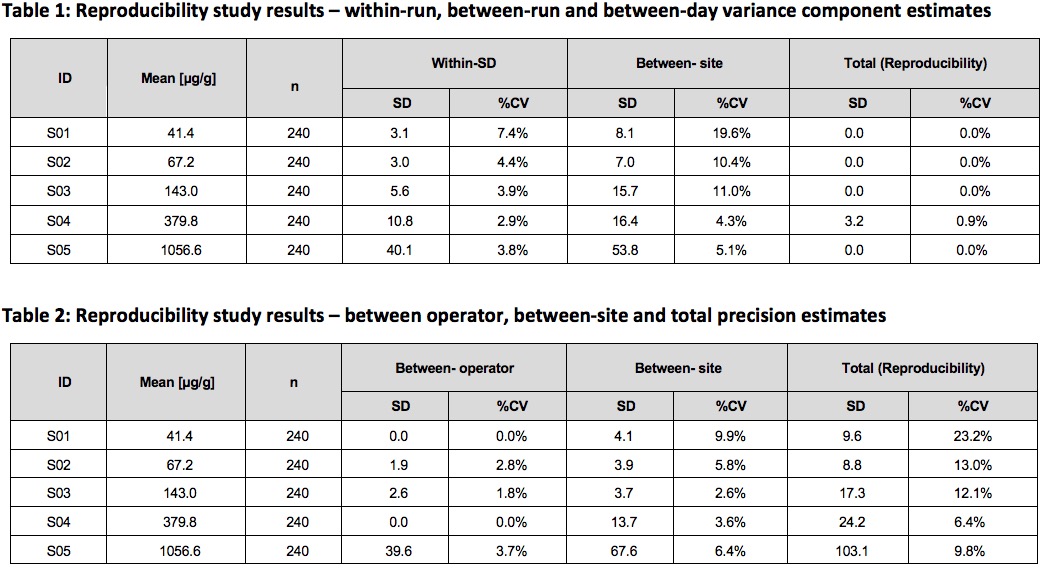
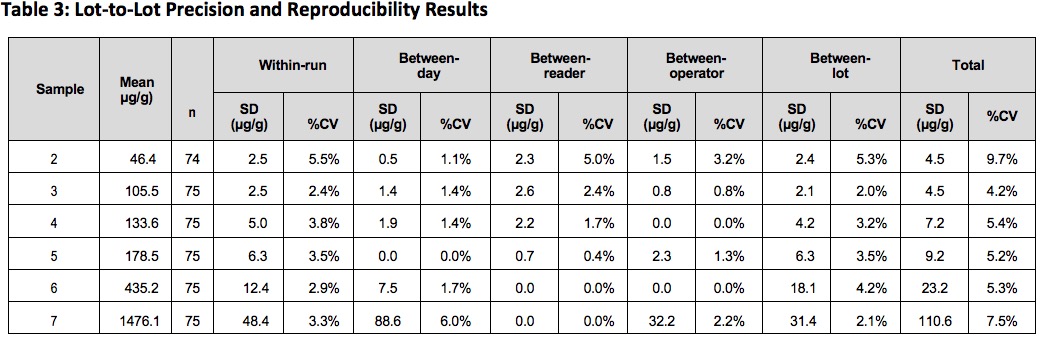
Precision and Reproducibility
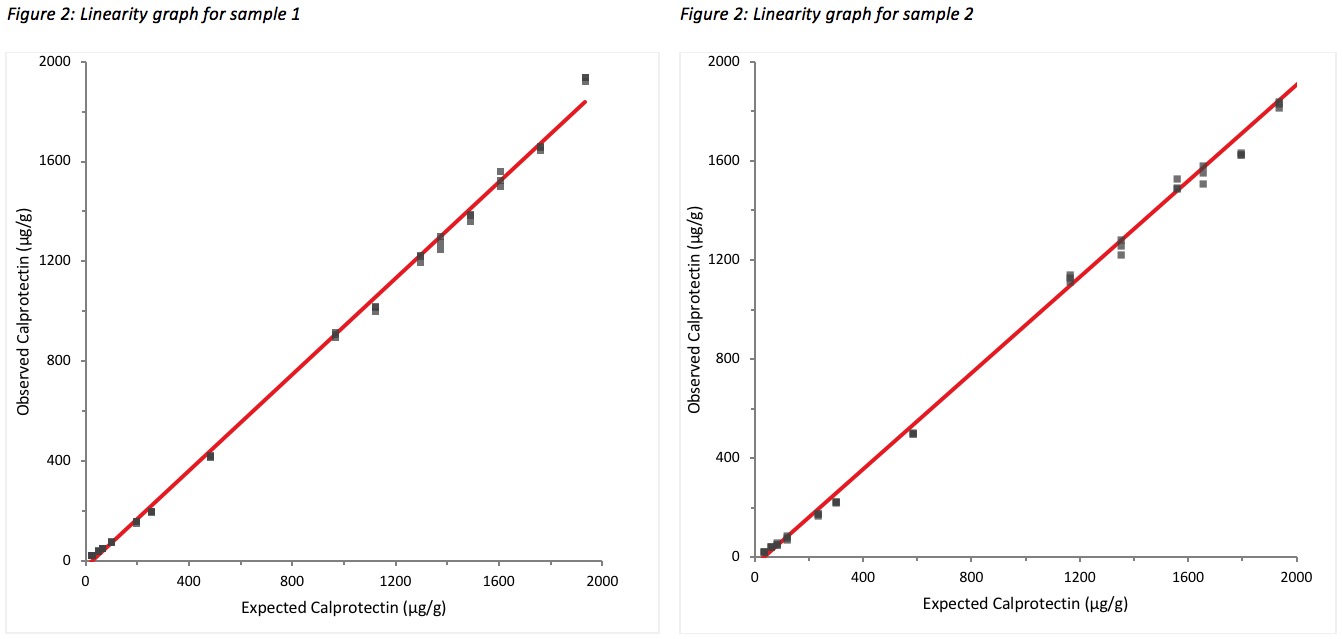
Linearity
 Download IFU and Catalog Numbers‡
Download IFU and Catalog Numbers‡
View the appropriate tab below and click on the blue underlined catalog number to view the Instructions for Use.
BÜHLMANN fCAL® ELISA – EK-CAL
ELISA Kit Sizes: EK-CAL: 96 wells (1 plate), EK-CAL2: 192 tests (2 plate)
BÜHLMANN Products are distributed in Canada by Inter Medico. For more information, email info@inter-medico.com or call 1.800.387.9643.
BÜHLMANN fCAL®ELISA – EK-CAL /CAL2
ELISA Kit Sizes: EK-CAL: 96 wells (1 plate), EK-CAL2: 192 tests (2 plate),EK-CAL2-WEX: 192 tests (without extraction buffer for use with CALEX®)
Extraction Device:
CALEX® Cap Device – B-CALEX-C50/ B-CALEX-C200/ B-CALEX-C500
Extraction Kit for BÜHLMANN fCAL® assays B-CAL-EX3-EX12




